-
-
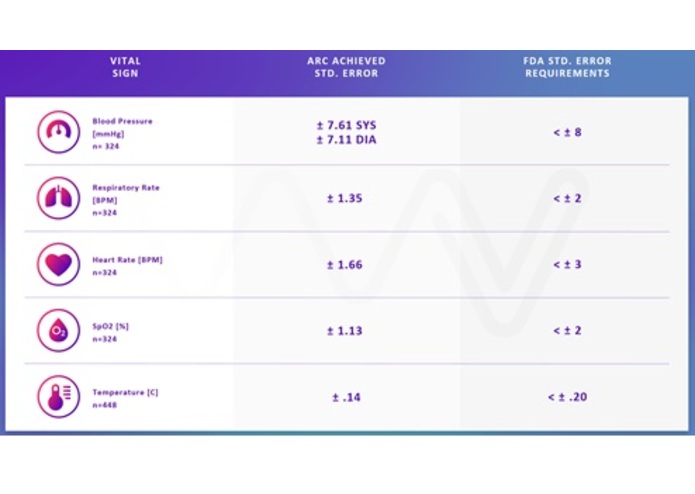
Figure 3. The VitalMVS device has gone through significant clinical trials ensuring clinical accuracy.
-
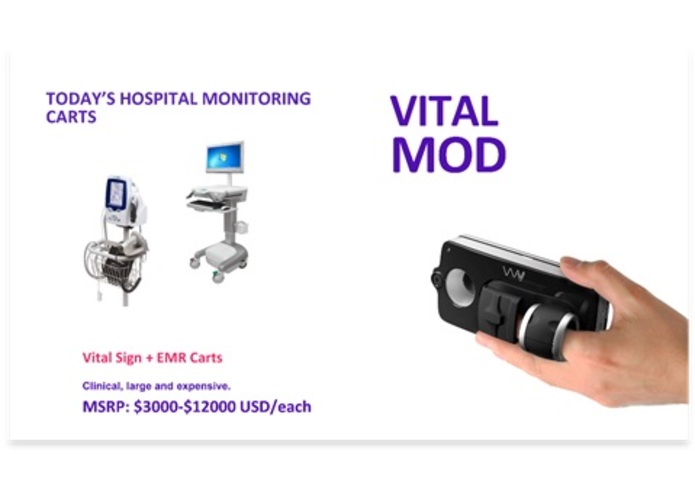
Figure 2. (Left) Traditional Vital Sign Cart vs (Right) Vital Health Mod..
-

Figure 1. The Vital Health measures blood pressure, heart rate, pulse oximetry, heart rate variability, and temperature in 3 minutes.
-

Figure 4. The Vital Glucose Sense measures non-invasive glucose. It measures heart rate, pulse oximetry, and HRV..
The mobile VitalMVS shown in Figure 1 will combine multiple vital signs measurement with transmission via Bluetooth to the VitalMVS Application and wireless (Bluetooth, WiFi®, or cellular) connectivity to the electronic medical/health record (EMR/EHR) from the point of care (PoC). The VitalMVS works in conjunction with the Vital App (Android and iOS). The Vital App sends the data to the Vital Cloud.
In the hospital environment, the VitalMVS is to replace the vital sign cart (~$2,500 to $12,000) currently used in hospitals (see Figure 2) for spot monitoring. The vital sign carts are costly and cumbersome to move around the hospital. The VitalMVS can measure the same vital signs as the cart and is estimated to be priced at $350. Even including a new tablet ~$80, the cost of the VitalMVS provides the same functionality including bar code scanning at a fraction of the cost. The VitalMVS sends the vital sign data to the Vital App through Bluetooth connectivity. The VitalMVS has a USB C interface for charging. The unit is compatible with all Android devices and will be compatible with all iOS devices (August 2018).
In the home environment, the integrated compact form factor of the VitalMVS in conjunction with the Vital App will guide the user through the workflow so that a trained nurse is not required.
The steps in the workflow are as follows:
A. Vital Measurement a. Turn on VitalMVS and open the Vital App. b. Follow the instructions on the screen and select what Vital you would like to measure. c. The patient inserts their finger into the device and waits for the system to perform a signal quality check to ensure that the finger is in the correct position (30 seconds). d. Measure Vital Signs i. Blood Pressure (SYS/DIA), heart rate, SpO2 – 1.5 minutes ii. Blood Pressure (SYS/DIA), heart rate, SpO2, and Resp. Rate – 3 minutes B. Measure Temperature a. Place the device 0.5 to 1 inch from the forehead and hold for 1 second to complete the measurement. C. View/Save Results. a. View the results and determine if they need to be saved. If saved, they will be automatically loaded into the cloud. b. Depending on the version of the app, once the data is successfully transmitted to the Vital Cloud, it will be sent to the EHR/EMR.
Figure 1. The VitalMVS measures blood pressure (systolic and diastolic), heart rate, pulse oximetry, heart rate variability, and temperature in 3 minutes.
Figure 2. (Left) Traditional Vital Sign Cart vs (Right) Smart VS.
Development
The VitalMVS captures vital signs through a combination of sensors. Temperature is calculated through the 510(k)-cleared InstaTemp MD infrared (K152905) sensor and proprietary body temperature algorithm. Blood pressure is calculated with an inflatable finger cuff and dynamic light scattering (DLS) sensor subsystem which measures blood flow after occlusion to calculate systolic and diastolic blood pressure. The photoplethysmogram (PPG) subsystem calculates heart rate and SpO2 from the ratio of 660 [nm] and 940 [nm] light absorption in the finger. Respiratory rate is calculated from a combination of the DLS and PPG subsystems. The captured and calculated values are transmitted via Bluetooth in real-time to a smartphone which displays the parameters. The core sensors and algorithms for these vital signs has been exclusively licensed from Elfi-Tech (http://elfi-tech.com/, Rehovot, Israel) Vital USA in conjunction with its outsourced engineering partner Avid Technologies (http://www.avid-tech.com/, Twinsburg, OH) have developed integrated Elfi-Tech’s sensors and algorithms and developed the mechanical and electrical design to comply with medical device performance (e.g. accuracy) and safety regulations. Vital USA is the exclusive holder of the IP associated with VitalMVS device.
Clinical Validation
The VitalMVS has gone through considerable clinical assessment and feasibility testing as shown in Figure 2. These trials performed in the USA, Canada, and Israel give Vital USA confidence that our clinical validation studies at Partners Connected Healthcare (Massachusetts General Hospital, Boston, MA) will be successful (see link http://www.iotevolutionhealth.com/topics/iotevolutionhealth/articles/436713-partners-connected-health-vital-usa-team-up-health.htm).
Figure 3. The VitalMVS device has gone through significant clinical trials ensuring clinical accuracy.
The clinical validation study at Partners Connected Healthcare will be the backbone of the 510k submittal for the VitalMVS. Validation will be performed against the existing standard of care equipment on n > 300 patients, across all clinically relevant ranges of vital signs.
What's next for Vital Health - Revolutionizing Patient Monitoring
The ability to monitor glucose non-invasively would change the lives of tens-of-millions of people worldwide. Vital USA has licensed the sensor and algorithms for non-invasive glucose measurement from Sanmina (Huntsville, AL). Included in the license from Sanmina is nitric oxide measurement, heart rate, pulse oximetry, and heart rate variability algorithms. The importance of nitric oxide is becoming more apparent as nitric oxide is a vasodilator, which expands the blood vessels, increasing blood flow and decreasing plaque growth and blood clotting.
Development
Vital USA will again work with Avid Technologies to improve the glucose and nitric oxide algorithms and sensor electronics to achieve clinical accuracy. The improvements make to the licensed technology are the sole intellectual property of Vital USA. The glucose sensors have already been built into the VitalMVS so when the algorithms are clinically validated, the algorithms can be seamlessly upgraded to the devices in the field. Furthermore, Vital USA is developing a standalone Glucose Finger Clip shown in Figure 4 which is very compact and supports measuring and monitoring of diabetes patients’ glucose, nitric oxide, heart rate, pulse oximetry, and heart rate variability levels.
Figure 4. The Vital Glucose Sense device measures non-invasive glucose through light scattering in 60 seconds. It simultaneously measures heart rate, pulse oximetry, and heart rate variability.






Log in or sign up for Devpost to join the conversation.