-
-
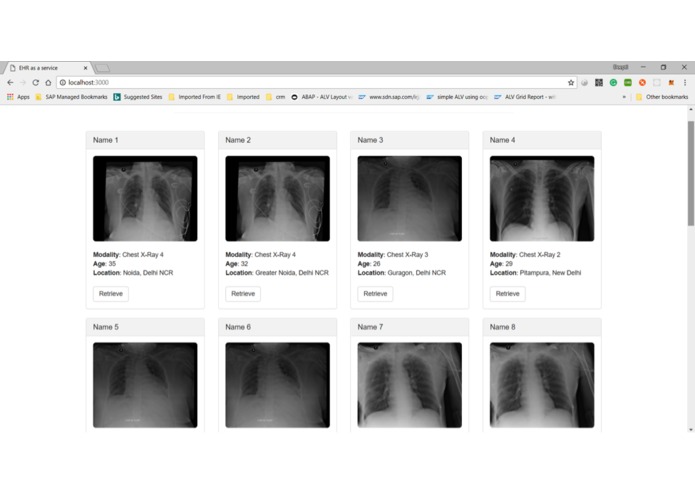
Radiology Query and Retrieve from Orthanc Pacs Interface and Scroll blockchain
-
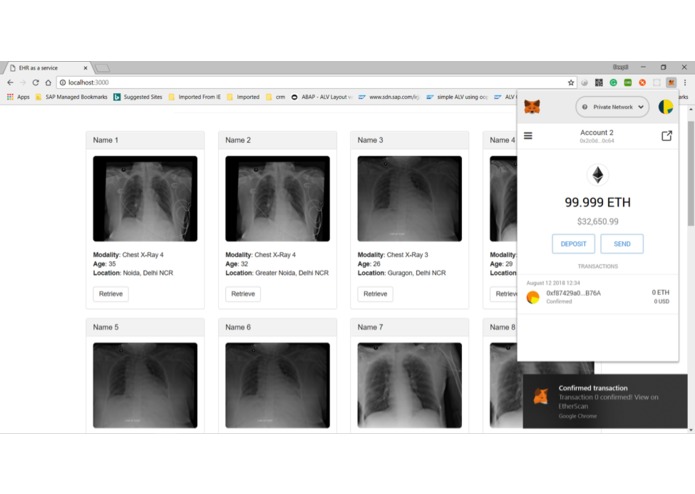
Successfuly Eth blockchain transaction on querying the EHR records
-
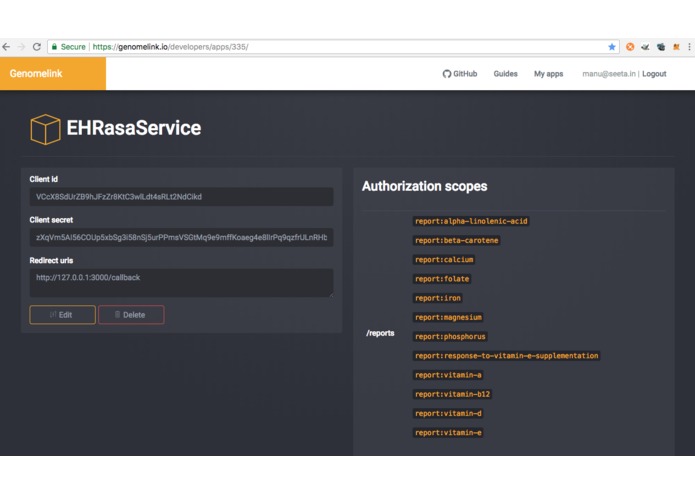
Connecting EHRs solution to GenomeLink API
-

Utilizing the GenomeLink API access rights and connecting the application after successful login
-
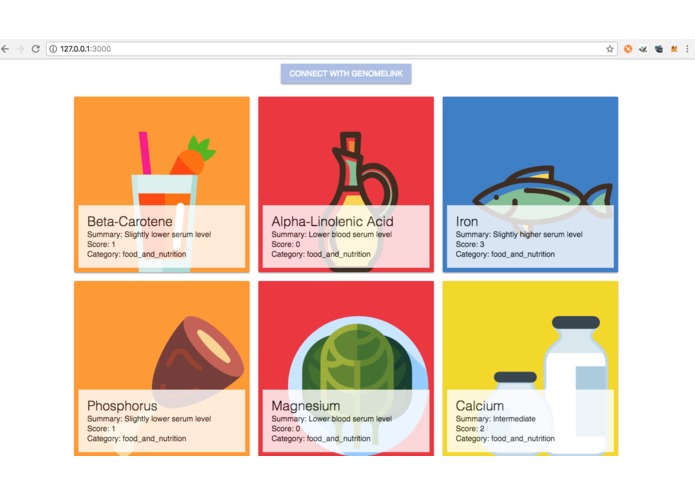
Nutrition Management according to EHRs medical report, prescribed medication and Genome understanding using GenomeLink API
-
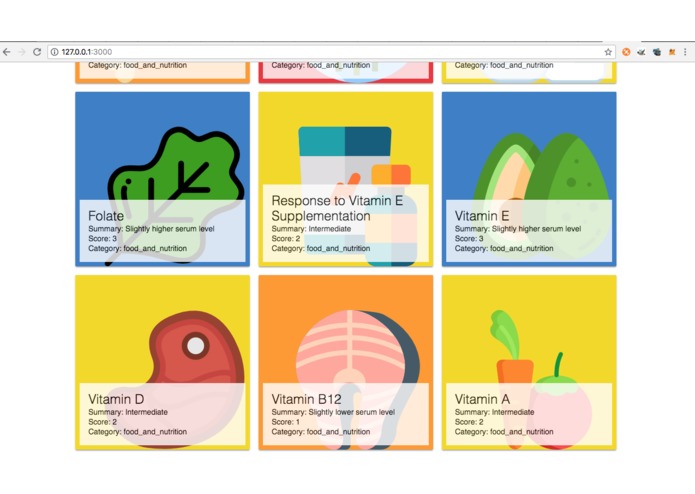
Determination of nutrients in food according to EHRs medical report, prescribed medication and Genome understanding using GenomeLink API
-
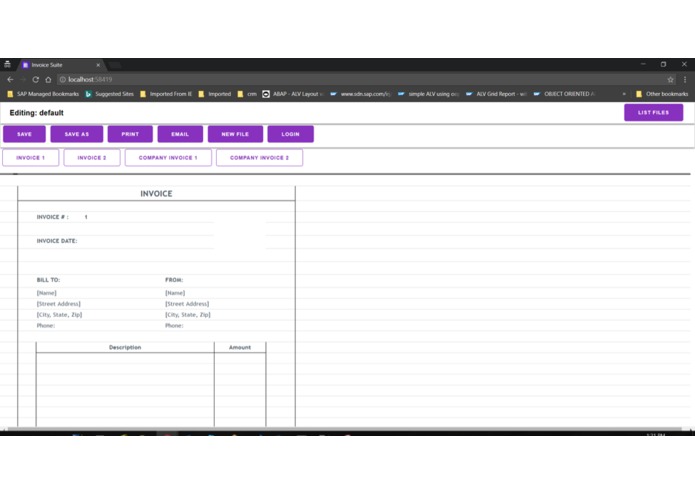
Medical Invoice app covering the prescribed medicines and dietary supplements for the patients
Inspiration
Institutional health records are not always open to other institutions, and institutional or personal health records are not always interoperable. These issues of care coordination are well known in healthcare, and are fundamental problems in medicine. Given its size and prominence, pharma and drug research organizations seeks to always be guarded against information security threats—from both external and internal actors. This means that they are required to sift through billions of events generated by network devices, endpoint solutions, and enterprise software. Collecting and aggregating EHR is a complex activity for drug design, development.
What it does
We are developing a technical solution using Filecoin/IPFS blockchain developer library along with Chainlink automation and Scroll blockchain network, open source spreadsheet engine to enable secure data storage, transparent data movement and data authenticity and borrowing funds for drug design and development using collaterals on Chainlink network. We enable pharma and drug design companies through EHR and Radiology services on the cloud. Further, we utilize a decentralize twitter application for health counselling, drug design and trials.
The solution needs to be maintained by regulators (e.g. MHRA, FDA), pharma and contract research organizations (CRO), which could be used in parallel with traditional clinical data management systems, framing the process as a transactional inter-organisational record keeping model between untrusted participants, thereby empowering global medical eco-system. Features:
• Just in Time Service: Availability of pharma companies medicine records across different stakeholder through secure blockchain network.
• Record Management: Quality documentation reduces the issues regarding testing procedures and standardization.
• Research: Enabling healthcare community by empowering pharma companies & the medical eco-system to do medicine trial testing securely, transparently.
• Data Security: Efficiently sharing of data (including personal data), privacy concerns and patient enrollment strategies.
• Transparency: Improving Data Transparency in Drug Testing Using Blockchain.
Explanation: Data Transparency : Availability of patient’s medical records across different stakeholder through secure Ethereum blockchain network and borrowing of Chainlink funds for drug design and development on XDC blockchain network. The platform utilizes Ethereum blockchain, IPFS via NFT.Storage, Nucypher i.e. patients and organizations who place their data on the exchange will be able to control which consortium entities have permission to access information.
Data Uniformity : Data is processed to make it uniform and stored in PACS (Picture Archiving Communication System) so that it can be utilized by different stakeholders on verified request. Also records are encrypted to avoid any tampering of the data over course of time.
Data Analytics : With the help of computer aided detection and machine learning algorithms, data can be further used for analysis and early prediction, drug discovery and development.
A greater and more seamless flow of information within a digital health care infrastructure, created by electronic health records (EHRs), encompasses and leverages digital progress and can transform the way care is delivered and compensated. EHRs helps in improved care coordination. EHRs helps in making health care ecosystem proactive, accessible and authentic. EHRs with the help of computer aided detection will help in early prediction and diagnosis of diseases.
We have developed our solution based on a variety of blockchain protocols and solutions and Chainlink blockchain network, which enables CROs to borrow funds for drug design and development.
CCIP NFT Deployment on Avalanche: https://drive.google.com/drive/folders/1txMlY2Z26xJOuEtyfC2F1Hhjq6_FDzqY
Demo Video of CCIP NFT Deployment on Avalanche: https://drive.google.com/drive/folders/1txMlY2Z26xJOuEtyfC2F1Hhjq6_FDzqY
Scroll Deployment (Hospital Role): https://sepolia.scrollscan.com/address/0x89E4F30AFB281689632535e1657D15243a83b802
Scroll Deployment (Patient/ Citizen Role): https://sepolia.scrollscan.com/address/0xDE16EdC78F1Af5865CCA458c8833069fDaf1Ca7b
Opensea minted NFT link (CCIP) : https://testnets.opensea.io/assets/sepolia/0x4c47dbeb2bec4e1bdb897430efed217c56d0270d/4
CCIP Explorer transaction Link: https://ccip.chain.link/msg/0xef336e431848d66424a1fb6db41a7a3560f4265c2adfa31055aa224817158633
CCIP Transaction hash link : https://ccip.chain.link/msg/0xf83f4d78c4cc86f9190319cc746eb7f0063a4740e1d78414a2b5fada8b104f80
Sucessfull ccip trasaction hash link ; https://ccip.chain.link/msg/0x3202aab91a2c818c515e9d03412942cc7832838e7b40754e046a100916001ad2
CCIP NFT fuji -> sepholia OpenSea link : https://testnets.opensea.io/assets/sepolia/0x4c47dbeb2bec4e1bdb897430efed217c56d0270d/0
Video screen cast of frontend and qr code scanning part and nft Minting : https://drive.google.com/drive/folders/1bkBGbHS_89y1nsEF5IMn79BBSNSDY8-S
How we built it
We developed the tool using ethereum blockchain network, store on IPFS/Filecoin via NFT.Storage, Chainlink for storing offchain metadata, Chainlink Data Feeds and automation for TPA claims and payments, NuCypher protocol for encryption, open source spreadsheet, open source PACS (Picture Archiving and Communication Systems) solution and Chainlink blockchain network for borrowing funds for drug design and development by CROs using collaterals.
Chainlink and Scroll blockchain network: Borrowing Chainlink, Scroll funds using collaterals for drug design research and development. Please visit: https://github.com/aspiringsecurity/EMTTR/tree/main/EHRs/xdc-borrow-funds
Chainlink DAO tracker: https://github.com/aspiringsecurity/EMTTR/tree/main/drugtestingworkflow/daos-tracker-tool
Chainlink Smart Contract Annotation Processor in Java: https://github.com/aspiringsecurity/EMTTR/tree/main/drugtestingworkflow/annotation-processor-java
Chainlink Visualization, Tabulation, Anaytics and Organization tool for user interaction at Uniswap, Sushiswap DEX exchange using OSS spreadsheet module with a wiki interface. Please visit https://github.com/aspiringsecurity/EMTTR/tree/main/Quotation-dapp-contract-management and https://github.com/aspiringsecurity/EMTTR/tree/main/Quotation-dapp-contract-management/sushiswap-analytics
Wish to extend the user analytics tooling with Dune like Analytics, collaboration with other DEX exchanges. Extensibility with Dune like Analytics tooling.
Improving data transparency during drug testing: We propose a Filecoin blockchain network maintained by regulators (e.g. MHRA, FDA), pharma and contract research organizations (CROs), to be used in parallel with traditional clinical data management systems (CDMS), framing the process as a transactional inter- organizational record keeping model between untrusted participants.
We are utilizing a variety of key Ethereum tools and libraries: metamask, truffle, embark, nvm, node.js, filecoin, IPFS, NFT.storage, chainlink datafeeds and automation, ethereum and a variety of blockchain protocols and systems along with AWS and XDC blockchain network. Our solution offers an Artificial Intelligence-based detection system that utilizes blockchain solutions for sorting information obtained from a variety of data management bodies, PACS systems deployed at monitoring spots.
A hierarchical arrangement of two core types of smart contract is required:
(i) A regulator contract, holding a data structure containing clinical trial authorization (CTA) details. This contract is owned and updated by regulators based on off-chain licensing agreements, and includes a container used to store trial contracts.
(ii) A trial contract, deployed by CROs using a function within the regulator contract, dependent on permission logic determined using the CTA data structure. Contains a data structure used to store the trial protocol, using IPFS8 or Ethereum’s native Swarm protocol where large file storage is required, with permission logic requiring protocol deposition and endpoint definition prior to the storage of subjects within a container. Subjects are added by CROs using a function within the trial contract, with permission logic restricting the calling of this function outside of the recruitment period defined in the protocol. The subject data structure contains anonymized subject information, consent documentation, and a container allowing storage of successive clinical measurements. Individual measurements are recorded, with full timestamping, in a format such as string- encoded JavaScript Object Notation (JSON), providing a flexible schema that can be adapted to any study type. Should data privacy be required, strings can be encrypted using public key encryption, with regulators holding a distinct private key for each trial con- tract, or using more elaborate techniques such as zero-knowledge proofs and homomorphic encryption as they become available.
Source code written in JavaScript and the Solidity smart contract programming language is provided under Data and software availability, allowing contracts to be implemented, and data to be written to and read from the blockchain. The scripts perform the following steps:
• Start JavaScript implementations of Ethereum and IPFS nodes, each connecting to local private networks.
• Deploy a regulator contract. A trial proposal, including proto- col documentation, is subsequently submitted to this contract by a CRO, with the documentation being stored using IPFS.
• If the proposal is accepted by the regulator, a trial contract is created. This contract is owned and administered by the CRO.
• Subjects are appended to the trial contract up until the trial start data. Synthetic data is then appended for each of the subjects, up until the trial end date.
• Finally, a script is provided to read all the data from the blockchain, providing a summary of each trial, and details of each subject and data points that have been added, with full timestamping.
Challenges we ran into
AWS deployment: We were using PV (paravirtual) based classic instance. Before August 15th, we were required to convert PV (paravirtual) instance to an HVM (Hardware Virtual Machine) and convert it from a classic instance to a VPC instance. We faced a number of challenges in converting our PV instance to an HVM instance as our instance was not reachable via ssh. We arrived at a good conclusion on the issue by doing the following: a. Created an ami of current classic instance. b. Launch a new instance from this ami after shifting to vpc as the base with new VPC security groups and updated volume. c. The newly launched instance is working and we are able to ssh and has the code base. We purchased the developer support plan using AWS credits and have being shared key pointers on converting our VPC based PV instance to a VPC based HVM instance. This challenge would not have been solved without the support of AWS team.
The third party administrators (TPAs) have to manually upload the medical history data to create the medi-claim/insurance based NFT. It is not a problem for a small set of patients where we can use csv to json conversion using open source libraries like phpexcel in ethercalc but problematic for larger number of patients. At this juncture, the TPAs have to manually connect their wallet and create the medi-claim/insurance claim NFT.
Accomplishments that we're proud of
A greater and more seamless flow of information within a digital drug discovery infrastructure, created by electronic medicine trial and test records as a service (EMTTRs), encompasses and leverages digital progress and can transform the way medicines are developed, tested and distributed to improve the global health economy and achievement of Sustainable Development Goals in Healthcare. Our blockchain solution, EHR as a service, can help in:
Legitimate storage of medical and environment records, medical invoices and environment data using open source PACS and IPFS for storage and react based spreadsheet application.
Records movement in a secure way using NuCypher Re-encryption Protocol, Query and Retrieve application on bootstrap and Orthanc DICOM open source solution.
Health counseling and environment awareness steps, preventive treatment and remediation over the ethereum blockchain network using a decentralized twitter application and Embark tools. EHRs can help in data cleansing, creating EHR, records legibility and coordination with hospitals, schools, pharmacies, diagnostic clinics, in case when movement of records is needed. The EHR application can be accessible using Internet browser, all the data is on the cloud.
What we learned
We can utilize SocialCalc, Machine Learning Models coupled with decentralized Chainlink, NFT.Storage, network tools, ethereum based infrastructure tools for analysis and prediction of diseases to provide early stage detection and prevention of medical compliance issues. We also witnessed the great eco-system available to developers to learn and contribute in the Ethereum and XDC blockchain eco-system.
What's next for EMTTR
We are evolving the solution in the following phases:
PHASE I – Requirements Analysis and Design (1 months)
Demonstrate the solution using spreadsheet and PACS software on cloud connected devices, Android phones, iPhones, first generation tablets. Procuring server hosting for storing and utilizing images and associated video data to prove that real time monitoring is viable. Set up timeline for planning the 2024 winter deployments and user training session Participate in community events organized by the incubator.
PHASE II (winter deployments and user training session) (8 months) -Create PoC based on designed specification. Complete the design of web interface.
Initial user testing, and POC refinement -Prototype Release- Start the pilot trials with 2-3 vendors. -Prototype Manufacturing at the Vendors location. -Prototype validation and assembly: Final Prototype assembly and validation; Refinements based on the mechanical prototype; Final Engineering CAD release. Manage and provide the hands-on task of exporting images and video reports from the customer to SEETA medical cloud system. Create and deploy a gateway service in the customer geo-location that will enable the continuing export of images and video reports to the SEETA medical cloud system. Completion of supporting collateral required to fulfil services and deliverables such as the equipment, supplies and other open source software tools. Continue the collection, data organization and management of images and associated video report data to improve computer aided detection using deep learning algorithms and integrate them with the platform. Survey on community’s needs, user interaction, selection of vendors and quality diagnostic centers where we could deploy full-scale pilot, possibly focused on mobile-platform organize a hardware agnostic program Enable pilot users to be developers of web based platform and contribute in improving the existing deep learning algorithms using websites like Kaggle. Focus on making the platform interoperable with a variety of vendor systems in different housing societies.
PHASE III (6 months)
Winter 2024-2025 deployment in hospitals, diagnostic labs, clinics, pharma companies and drug design and development institutions.
Built With
- blockchain
- ethercalc
- genomelink-api
- html
- javascript
- pacs
- php
- python
- shell








Log in or sign up for Devpost to join the conversation.