-
-

CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-

CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-

CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-

CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-

CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-

CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-

CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-
CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-

CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-
CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-

CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-

CORSVP COVID-19 Registry Screening Volunteer Platform (CORSVP™)
-
Large U.S. covid-19 vaccine trials are halfway enrolled, but lag on participant diversity, Washington Post, August 27, 2020
Inspiration
Hundreds of thousands of volunteers are sought to register for clinical trials to test for a vaccine to COVID-19. Oracle developed a cloud service called the COVID-19 Prevention Trials Network (CoVPN) Volunteer Screening Registry. This service is designed to allow people to volunteer for clinical trials, and assists clinical trial sites to choose participants based on trial-defined demographics. The issue is the application is web-based and doesn't utilize the sensors on a mobile device.
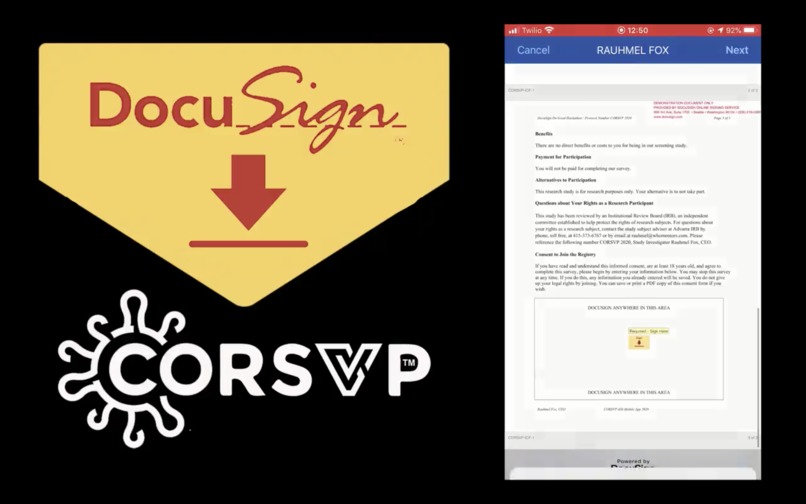
Title 45 CFR § 46.117 requires informed consent shall be documented by the use of a written informed consent form approved by the IRB and signed (including in an electronic format) by the subject or the subject's legally authorized representative. A written copy shall be given to the person signing the informed consent form.
This can be accomplished using the DocuSign eSignature API combined with Apple ResearchKit framework.
What it does
The registry is a platform for collecting basic information for screening for being invited to a clinical trial. A PDF copy of informed consent form stating that the elements of informed consent required can be easily completed and retained for records. After consent is obtained, ResearchKit framework permits the collection of data from the mobile device.
How we built it
Followign DocuSign iOS SDK and integrating ResearchKit
Starting with the Docusign, the investigator can read the consent form or send it to the volunteer. Or the individual can sign on their own.















Log in or sign up for Devpost to join the conversation.