30.4.2020 Update and Acknowledgment
CoroVent was selected among the challenge winners in the EUvsVirus Hackathon put on by the European Commission this past weekend, competing in the Respirator & Ventilators category.
The competition led by the European Innovation Council in close collaboration with the EU member states had over 9000 participants with 2163 competing projects in 37 categories.
One of the benefits of finishing among the best is the chance to participate in an upcoming "Matchathon" - matchmaking weekend bringing together winners, partners and potential investors.
This was gratifying to the CoroVent team to have recognition on an EU-wide scale, especially as we go into early internal production Sunday, and launch our full production of tested units for the public next week.
Why CoroVent
We connected biomedical engineers with 25 years experience in Ventilator design together with Covid ICU doctors and manufacturing experts to develop in 6 weeks professional Ventilators optimized for protective care of Covid-19 patients with the most severe respiratory failures, satisfying all EU requirements to allow production of 500+ units per day starting from may.
Inspiration and motivation
When the Coronavirus pandemic hit Italy, we were terrified by reports that doctors had to decide who got a ventilator (and so would get a chance to live) and who didn’t, all because of a ventilator shortage.
Analysts predict that 880,000 more ventilators will be needed globally to cope with the coronavirus outbreak. We knew that this would soon affect other EU countries, and so we would have to make our own.
On our initial call with Covid ICU doctors and biomedical engineers it became obvious that hacks based on bag-valve masks (Ambu bags) are useless for Covid patients, as sufferers require a very sophisticated ventilator (see explanation video)
All such ventilators on the market were too complex and not possible to make in large quantities (as they use unique components). We decided we needed a scaled-down version to the single mode best suited for Covid patients.
But there was no such simplified project anywhere, so we had to start from scratch. And since the pandemic created a rush on all related medical supplies, we decided to use components commonly employed in other industries rather than fight the demand.
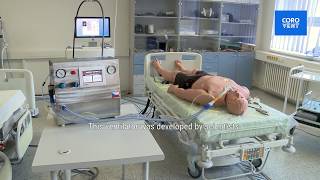
What CoroVent does
CoroVent presentation (5:49) for ICU professionals
In accordance with design principles for protective ventilation, all of CoroVent’s basic ventilation parameters can be adjusted. There is currently one single ventilating mode implemented that was selected by Covid doctors as the best for their patients - volume-controlled pressure-limited ventilation. It is adjusted via a touch screen interface, specially designed with input from ICU doctors so they could easily understand it without need for extra training. Some other modes will be added later via software update, as soon as we verify their flawless operation.
Design
Scheme, technical description and key custom-made components are publicly documented and open sourced . The design requirements as previously set out were (in no particular order):
- No single point of failure
- Connection to hospital oxygen and air medicinal gas systems, or operation with gas cylinders
- Electronic control system
- Multilingual user interface via touch screen display similar to those frequently used with medical ventilators
- Alarm signalization to flag inadequate ventilation, gas pressures, etc.
- Integrated power backup system (currently ~ 180 minutes of operation)
- Rugged design suitable for use in temporary facilities
- Increased resistance to various disinfectants and liquids, both surface & penetration (IP21 test)
- Adherence to the latest international guidelines, such as the UK released instructions, was taken into consideration.
Parameters
- Dimensions: 50 x 45 X 21 cm (w x h x d), weight 25kg
- Power requirements (EU version) 100 V – 240 V / 50 – 60 Hz
Controlled/adjustable parameters:
- VT volume (150-800 ml)
- inspiration/expiration time ratio I:E 1:1 - 1:3,
- respiratory frequency RR 10-30/min,
- positive end expiratory pressure PEEP (0-30 cmH2O),
- maximum inspiratory pressure Pmax (up to 65 cmH2O),
- inspiration limit pressure Plim (15–45 cmH2O),
- Oxygen inspiratory fraction (FiO2) 21–100 %
Measured parameters - Inspiratory volume VTi (ml), expiratory volume VTe (ml), inspiration/expiration time ratio, maximum inspiratory pressure Pmax (cmH2O), positive end expiratory pressure PEEP (cmH2O), ratio of inspiration time to total respiration cycle time Ti/T
How We Built the CoroVent
Our core team consists of 30+ members from over 20 companies and institutes - prof. Ing. Karel Roubík, Ph.D., his assistants and doctoral students from the Faculty of Biomedical Engineering of Czech Technical University in Prague with over 25 years specializing in medical ventilation, doctors from ICU units, Czech and EU companies with huge experience in fast development and production of pneumatic/electrical components, together with other experts in logistics, project management, funding, medical certification etc. Read the whole story as published in highly respected business journal “Hospodářské noviny”.
From day one we all focused entirely on this project only, investing together over 35,000 hours of work to make it happen. We knew that the main issues were not about the ventilator itself (actually we had its design done in 1 week and the first working prototype the week after), but more the ability to manufacture it in large quantities and get all the necessary official tests to validate for non-medical device use in hospitals during this crisis situation.
The ventilator is assembled in a region with one of the highest unemployment rates in the Czech Republic by Dawell company, part of the MICo Group, that was a key part of our team. The design is made to resemble LEGO toys, easily assembled from building block components by trained workers in less than an hour, with logistics and manufacturing processes optimized by professionals. We have also signed an agreement with 3 other neighboring assembly plants used for production of automotive parts that had to close the operation because of Covid19 crisis. We also focused on the very detailed testing of every unit, which takes up to 24 hours. Everything is subordinate to the key constraints: we need ventilators fast, but also need them completely safe for use with Covid patients, same as would be for the best ventilators on the market.
100% of our components are produced in EU/European countries
- MICo Group (Czech Republic)
- Beckhoff (Germany)
- Festo (Germany)
- Puls (Czech Republic, Austria, Poland)
- and other companies from Austria, Switzerland, Sweden, Italy and Spain
Challenges We Ran Into
Our biggest hurdle was to find non-medical components that would work in a medical ventilator. E.g. standard valves would be tested over millions of cycles in a medical setting, which would make them nonfunctional after a few weeks of continuous in-and-out breathing cycles. They also exhibit lags and other behavior that is perfectly OK for use in cars and unrelated scenarios, but would kill the patient if used in a ventilator. We had to test tens of different models and brands until we found the ones that work as needed under proper constraints, or even had to persuade the manufacturers to make a special version for us.
The second challenge was to get enough components for planned units - we are not a big brand like Dyson or Tesla, and have not received any funding yet from CZ, EU or US, so it’s difficult to purchase enough up front. Several times it happened that we wanted some part that had up to a half million units in stock, but the following day none were left, as someone from General Motors came and bought them all up just in case they might need some for their ventilators later on.
This is an issue we believe the hackathon might address with the EU that could help us and others - that the EU should give priority to and protect projects like CoroVent that have specific social need along with proven design and working prototypes - by supporting critical component production and limiting exports from the EU as non-EU countries have been known to do.
Accomplishments that we’re proud of
- Passing all the tests in official testing laboratory required by CZ and EU (see test report)
- Great acclaim and support by the Czech public as our initial crowdfunding campaign for 10 MIO CZK was exceeded by 40 % in just 18 hours
- Fast development of 6 weeks as the project went from concept to design to 1st prototypes, test & production
- Rapid team integration of relative unknowns from different sectors
- Positive feedback from Czech government, including the Prime Minister
- Continuous positive Media coverage, including
What We learned
- Medical parts will be exhausted in an epidemic
- More generic parts are available, but often not up to medical demands or standards
- Funding is less important than having in-country allies to shepherd through the acceptance process
- Open specs can lead to shortages of specified parts, often leading to fewer overall machines produced and thus lives saved
What's next for CoroVent
- Certification and/or usage exemptions in multiple additional regions, starting with other EU countries
- Scale up the demand so we can fit the production capacity of 500+ pcs a day
- Enhanced training of hospital personnel using short instruction video, et al. (even though design is largely intuitive)
- UN support as well as targeted partnership with government entities
- Eventually identify suitable parts alternatives to ensure supply
Price, funding, expenditure, business plan
- current team time investment together 35.000 hours in last 6 weeks
- costs for setting up the production line, logistics etc.: 1 MIO €
- cost of material already on stock or paid in advance (necessary for some parts we needed): 3 MIO €
- crowdfunded 100 ventilators for hospitals in the Czech Republic (0.5 MIO € in less than 1 day)
- other funding - not yet (all expenditures were covered by team members themselves without outside support).
- production capacity - 100+ pcs/day with one shift, 300+ pcs/day in current facility, 3 other neighbor facilities signed on for a total maximum capacity of 850+ pcs per day
- service costs, etc. - should be covered by hospital contracts with their service organizations, we will take care of training, spare parts etc.
- current bare material cost per one unit: ~6K € (price of some parts grew 4 times or more over the last 6 weeks due to high demand)
- calculated price (will vary according to demand, EU/national support and requirements from other countries), probably somewhere between 8000-10,000 €.
Current competition on the market
There is very limited competition as existing ventilators that can handle intubated patients with Covid typically cost $25,000 and $50,000 (e.g. here) but the demand is so high that you cannot buy them. Also because of their complexity it is impossible to produce them in large quantities. E.g. Slovak ventilator producer Chirana a month ago promised only 300 ventilators for Slovakia - by the end of June.
There are many other teams that wanted to produce usable ventilators, but they largely failed (e.g. Dyson), found that they chose an unsuitable design for Covid patients (e.g. Valeo France) or are still not up to production stage as compared to our CoroVent project.
We can save millions of lives
If you compare Covid mortality rate in countries where they got into trouble with ICU and ventilator capacity (Spain 10.25%, Italy 13.51%, United Kingdom 13.69%) to those lucky ones who were better prepared or set-up stricter restrictions (Germany 3.66%, Czech Republic 2.97%), you can see a tremendous difference.
Today (26.4.2020) we have 1.140 MIO confirmed infections in European countries (without Russia) and 119,000 confirmed deaths out of 620 MIO people. Even if we would determine that 50% of infected people were not traced, it means that fewer than 0.4% got through the illness. If it is true that 70% of the European population could be infected in the next few years, we are still just scratching the surface. If we get our ICU’s better prepared and equipped with enough ventilators, we would lose “only” those 3% of those 70% of our 620 milions of citizens, not 13%.
And of course a similar situation exists outside Europe and we must help these as well if we want to help the world breathe again and restart its economies. So far CoroVent is the only viable project of this sort that could be produced quickly in large quantities, so that it will be in the hospitals when the new waves of pandemics arrive. And the EU will take the credit, if you will help us scale the production.
How EU and partners/organizers of this hackathon can help us
- proper funding or orders could help us scale up production faster to decrease the price
- help us be recognized by national authorities, hospitals, Ministries of Health… so that we can get their approval to use our ventilators in their hospitals and get their orders
- help to get more national support for our component vendors in various EU countries, so that we can deliver more units faster
Built With
- blood
- sweat
- tears








Log in or sign up for Devpost to join the conversation.